AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Capto s chemical compatability11/30/2023  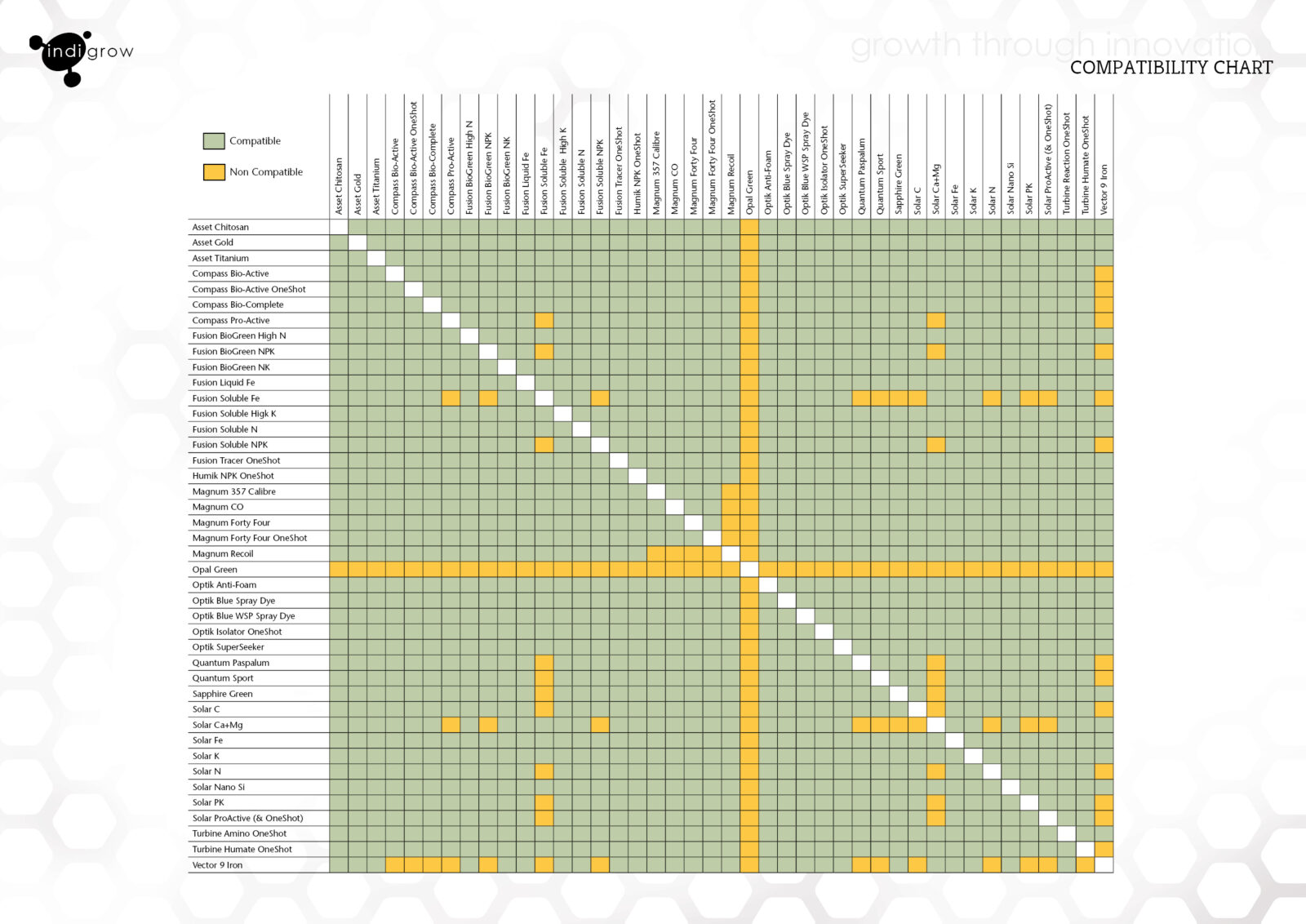
In anion exchange chromatography, the exchanging ions are anions and the equation is represented as follow In this process the cation M + of the eluent replaced with the analyte cation C + bound to the anion X - which is fixed on the surface of the chromatographic support (S). Assuming that the exchanging ions (analytes and ions in the mobile phase) are cations, the competition can be explained using the following equation The analyte ions and similarly charged ions of the eluent compete to bind to the oppositely charged ionic functional group on the surface of the stationary phase. 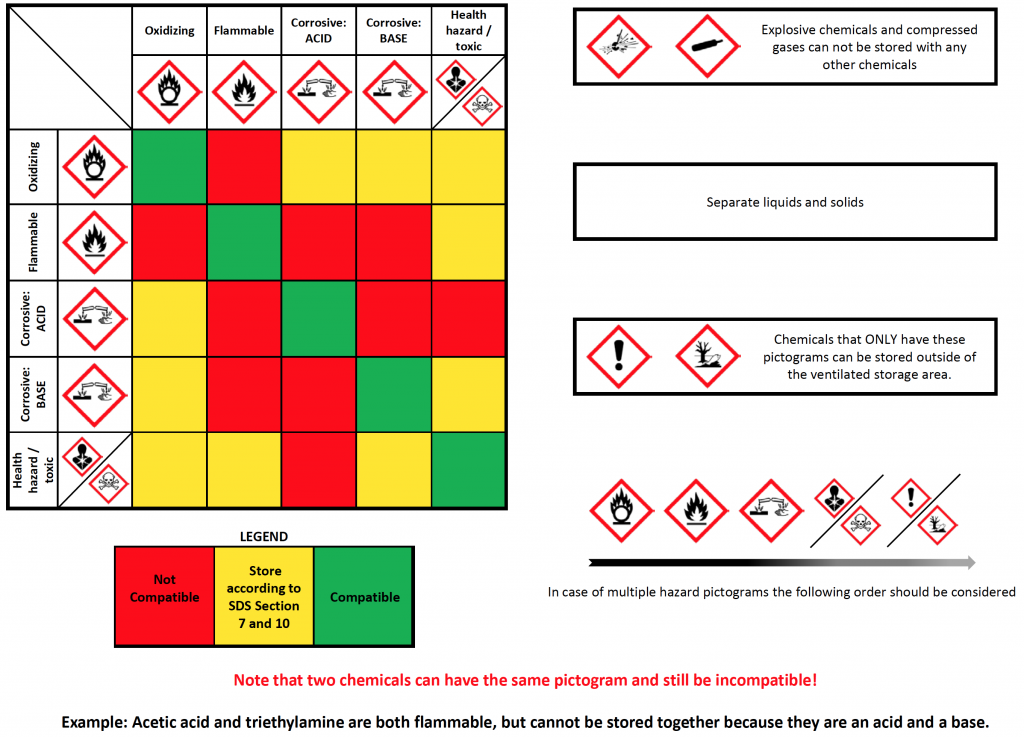
Ion exchange chromatography involves separation of ionic and polar analytes using chromatographic supports derivatized with ionic functional groups that have charges opposite that of the analyte ions. Separation based on the binding of analytes to positively or negatively charged groups which are fixed on a stationary phase and which are in equilibrium with free counter ions in the mobile phase according to differences in their net surface charge ( Figure 1). Cations are separated on cation-exchange resin column and anions on an anion exchange resin column. Exchangeable matrix counter ions may include protons (H +), hydroxide groups (OH -), single charged mono atomic ions (Na +, K +, Cl -), double charged mono atomic ions (Ca 2+, Mg 2+), and polyatomic inorganic ions (SO 4 2-, PO 4 3-) as well as organic bases (NR 2H +) and acids (COO -). Ions which exist in a state of equilibrium between the mobile phase and stationary phases giving rise to two possible formats, anion and cation exchange are referred to as counter ion ( Figure 1). 
The stationary phase usually made from inert organic matrix chemically derivative with ionizable functional groups (fixed ions) which carry displaceable oppositely charged ion. Mobil phases consist an aqueous buffer system into which the mixture to be resolved. Ion-exchange chromatography which is designed specifically for the separation of differently charged or ionizable compounds comprises from mobile and stationary phases similar to other forms of column based liquid chromatography techniques. The popularity of ion exchange chromatography has been increased in recent years because this technique allows analysis of wide range of molecules in pharmaceutical, biotechnology, environmental, agricultural and other industries. Ion-exchange chromatography has been used for many years to separate various ionic compounds cations and anions and still continues to be used. published a method for anion chromatography in 1979 and this was followed by a similar method for cation chromatography in 1980. In modern form ion exchange chromatography was introduced by Small, Stevens and Bauman in 1975. In order to separate proteins an ion exchange chromatographic method was reported by Peterson and Sober in 1956. 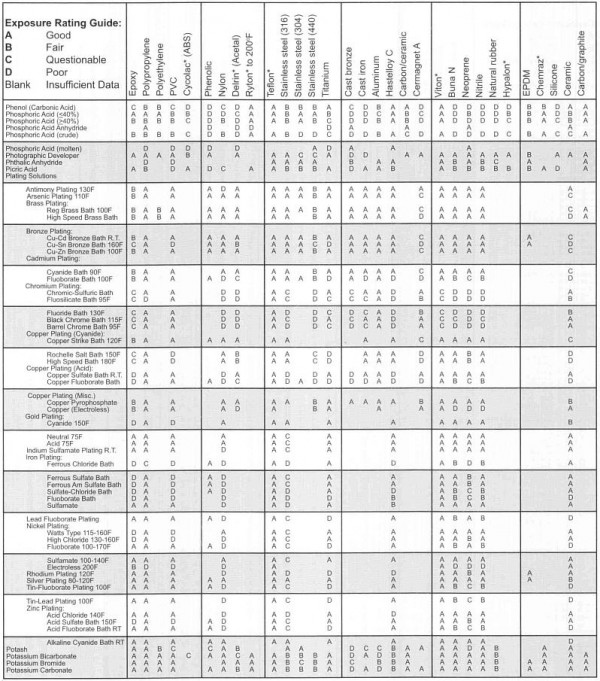
Beginning in the 1950s, Kraus and Nelson reported numerous analytical methods which are used for metal ions based on separation of their chloride, fluoride, nitrate or sulfate complexes by anion chromatography. Spedding and Powell published a series of papers describing practical methods for preparative separation of the rare earths by displacement ion-exchange chromatography in 1947. The earliest report of ion-exchange chromatography date back to 1850, Thompson studied the adsorption of ammonium ions to soils. Biomolecules display different degrees of interaction with charged chromatography media due to their varying charge properties. The separation is based on the formation of ionic bonds between the charged groups of biomolecules and an ion-exchange gel/support carrying the opposite charge. This chromatography is one of the most important adsorption techniques used in the separation of peptides, proteins, nucleic acids and related biopolymers which are charged molecules in different molecular sizes and molecular nature. Ion exchange has been the predominant form of ion chromatography to date. Two distinct mechanisms as follows ion exchange due to competitive ionic binding (attraction) and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support, play a role in the separation in ion chromatography. Ion chromatography separation is based on ionic (or electrostatic) interactions between ionic and polar analytes, ions present in the eluent and ionic functional groups fixed to the chromatographic support. Ion-exchange chromatography (IEC) is part of ion chromatography which is an important analytical technique for the separation and determination of ionic compounds, together with ion-partition/interaction and ion-exclusion chromatography. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
